A meta-research study revealed several challenges in obtaining placebos for investigator-initiated drug trials - ScienceDirect
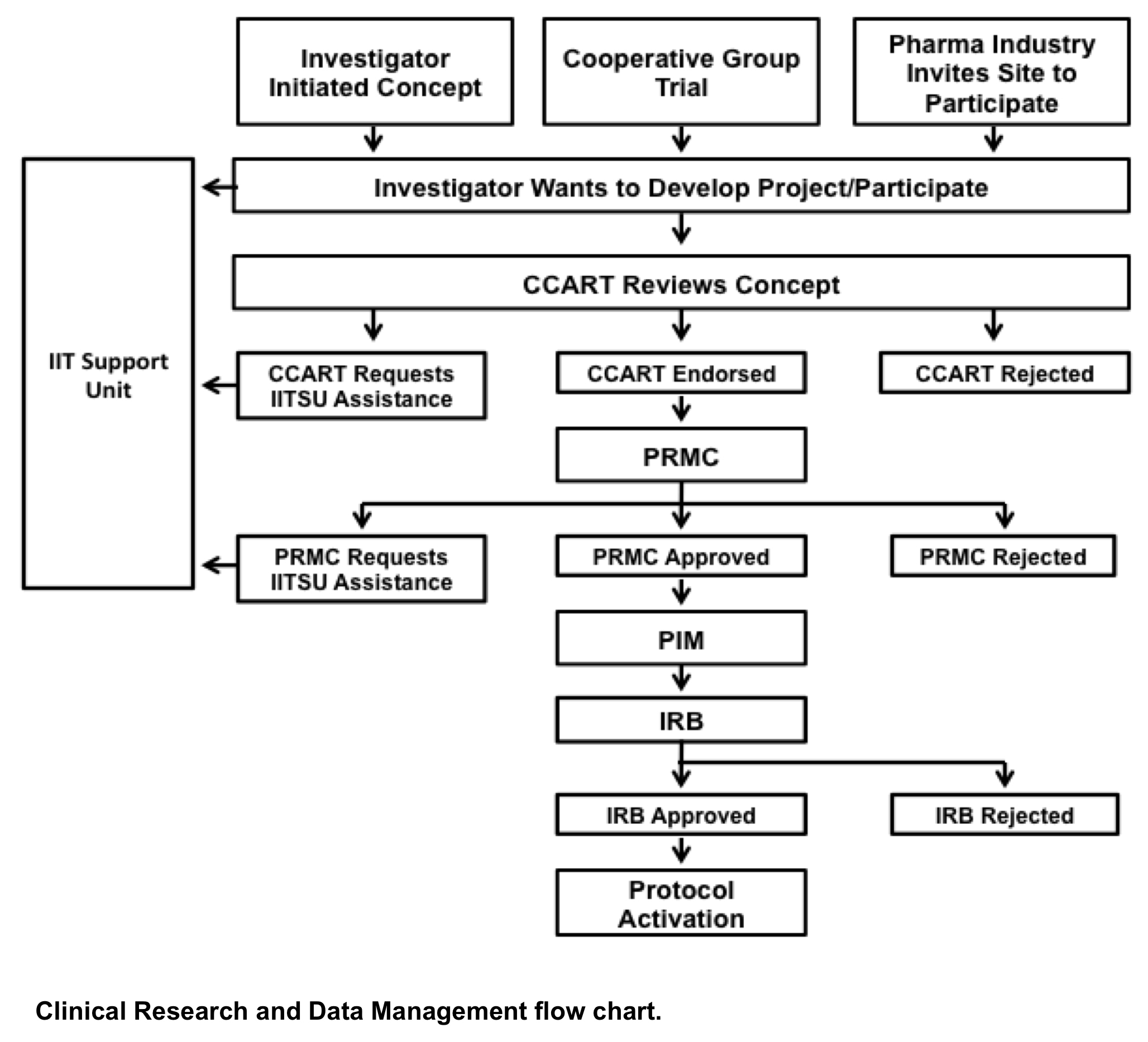
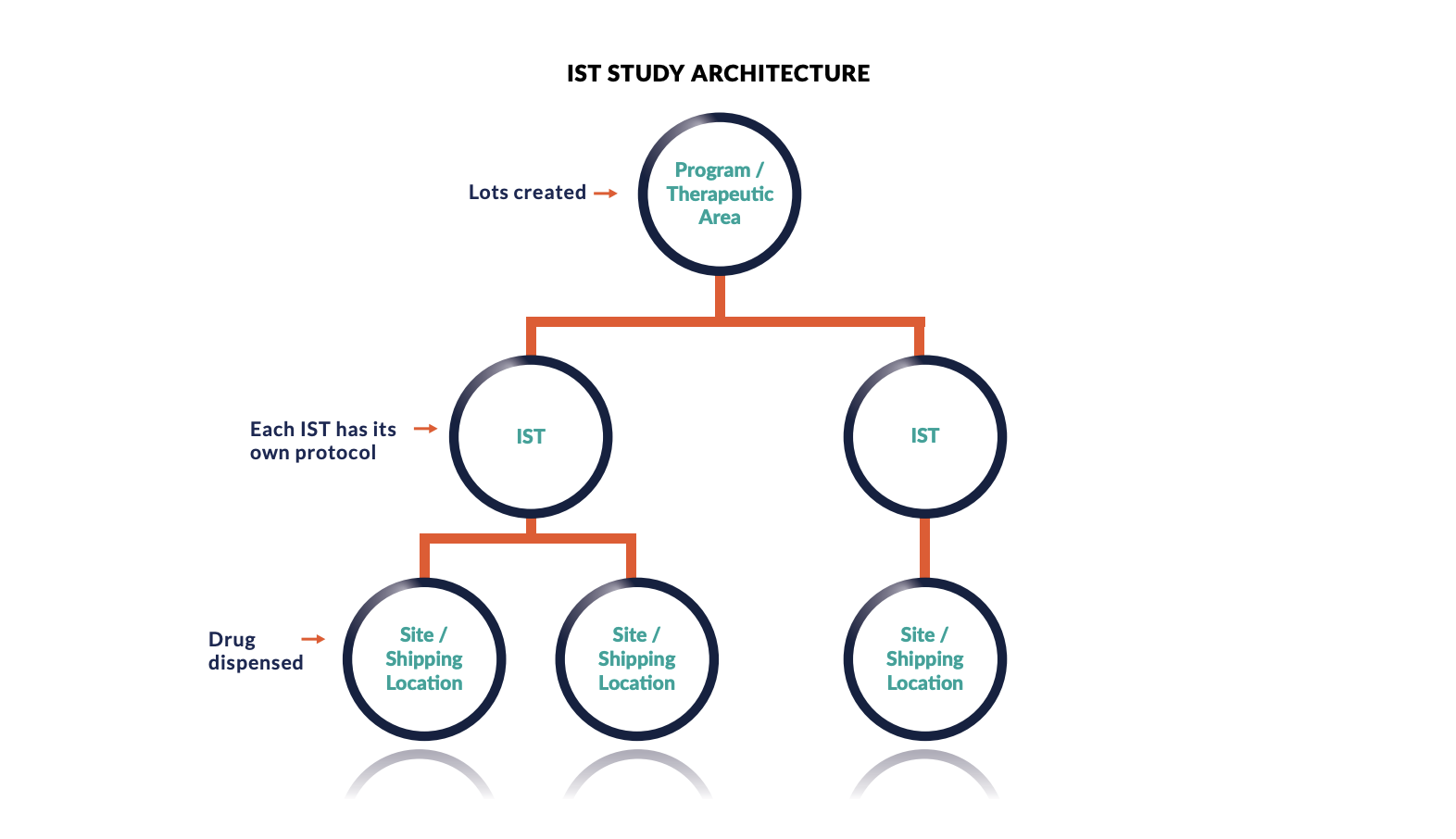
Investigator Initiated Trials (IIT) – Considerations and Guidance from the Perspective of Clinical Trial Supplies and GMP | ISPE | International Society for Pharmaceutical Engineering

Industry Viewpoints: Investigator-Initiated Studies – A Challenge for Supply with IMPs? - Clinical Trials Arena

MJA on Twitter: "Value proposition of investigator‐initiated clinical trials conducted by networks … #freeaccess 1 week … "Investigator‐initiated trials run by clinical trial networks provide net economic benefits to health systems" https://t.co ...

Guide for Investigator Initiated Trials: 9783805596848: Medicine & Health Science Books @ Amazon.com