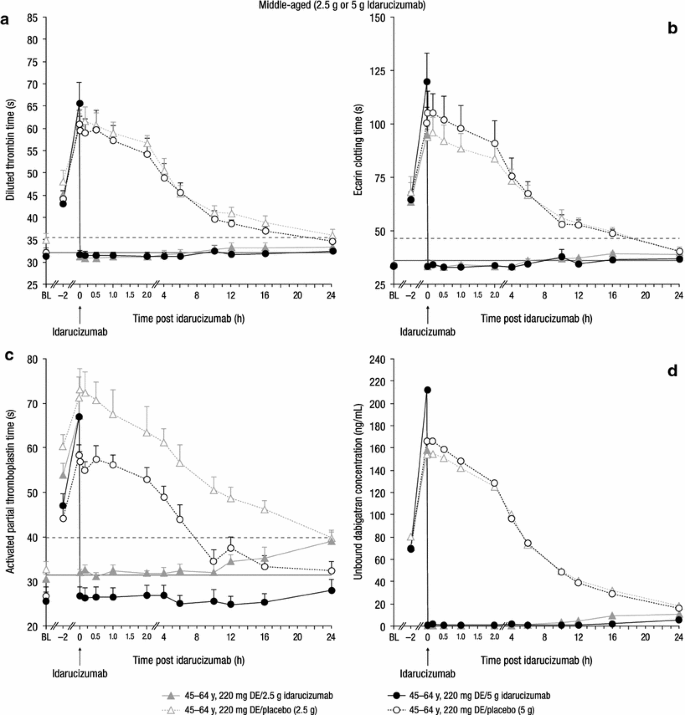
Effect of Age and Renal Function on Idarucizumab Pharmacokinetics and Idarucizumab-Mediated Reversal of Dabigatran Anticoagulant Activity in a Randomized, Double-Blind, Crossover Phase Ib Study | SpringerLink

Dabigatran Reversal With Idarucizumab in Patients With Renal Impairment | Journal of the American College of Cardiology

Safety, tolerability, and efficacy of idarucizumab for the reversal of the anticoagulant effect of dabigatran in healthy male volunteers: a randomised, placebo-controlled, double-blind phase 1 trial - The Lancet

Idarucizumab, a Specific Reversal Agent for Dabigatran: Mode of Action, Pharmacokinetics and Pharmacodynamics, and Safety and Efficacy in Phase 1 Subjects - ScienceDirect

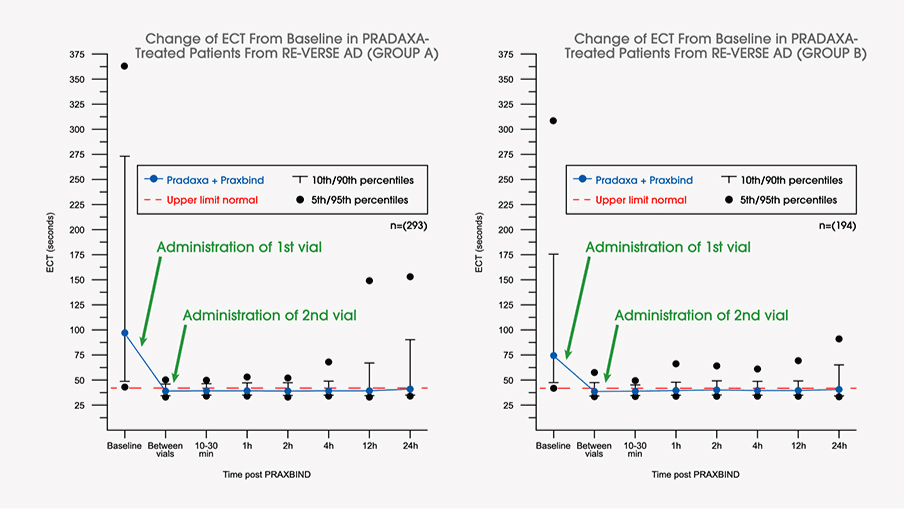
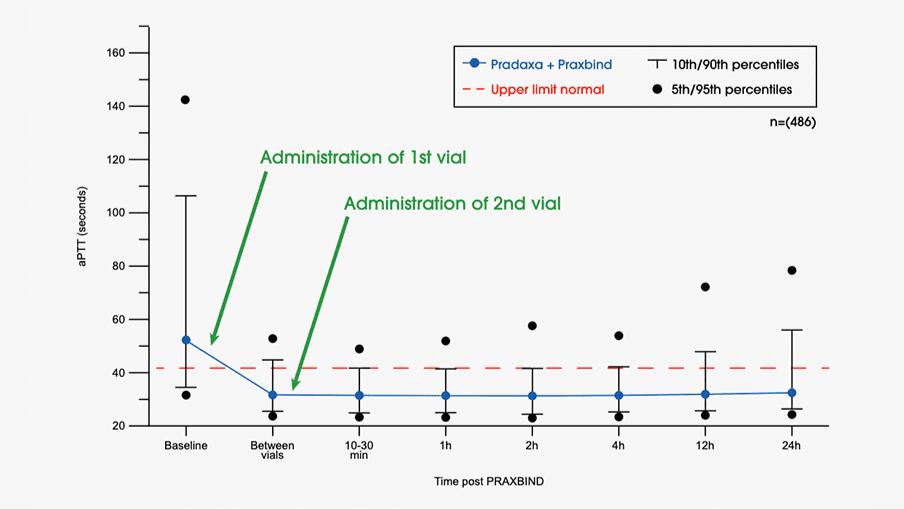
These highlights do not include all the information needed to use PRAXBIND safely and effectively. See full prescribing information for PRAXBIND. PRAXBIND® (idarucizumab) injection, for intravenous useInitial U.S. Approval: 2015
Idarucizumab, a Specific Reversal Agent for Dabigatran: Mode of Action, Pharmacokinetics and Pharmacodynamics, and Safety and Ef

Safety, tolerability, and efficacy of idarucizumab for the reversal of the anticoagulant effect of dabigatran in healthy male volunteers: a randomised, placebo-controlled, double-blind phase 1 trial - The Lancet
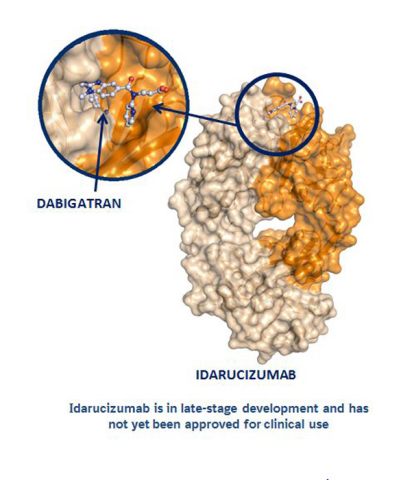
Boehringer Ingelheim submits applications for approval of Idarucizumab, an anti-anticoagulant - Labiotech.eu
Interpretation of idarucizumab clinical trial data based on spontaneous reports of dabigatran adverse effects in the French phar

PDF) Effectiveness and Safety of Dabigatran Reversal with Idarucizumab in the Taiwanese Population: A Comparison Based on Eligibility for Inclusion in Clinical Trials
Clinical Effectiveness of Idarucizumab in Dabigatran Reversal | Wang | Journal of Cardiology and Therapy
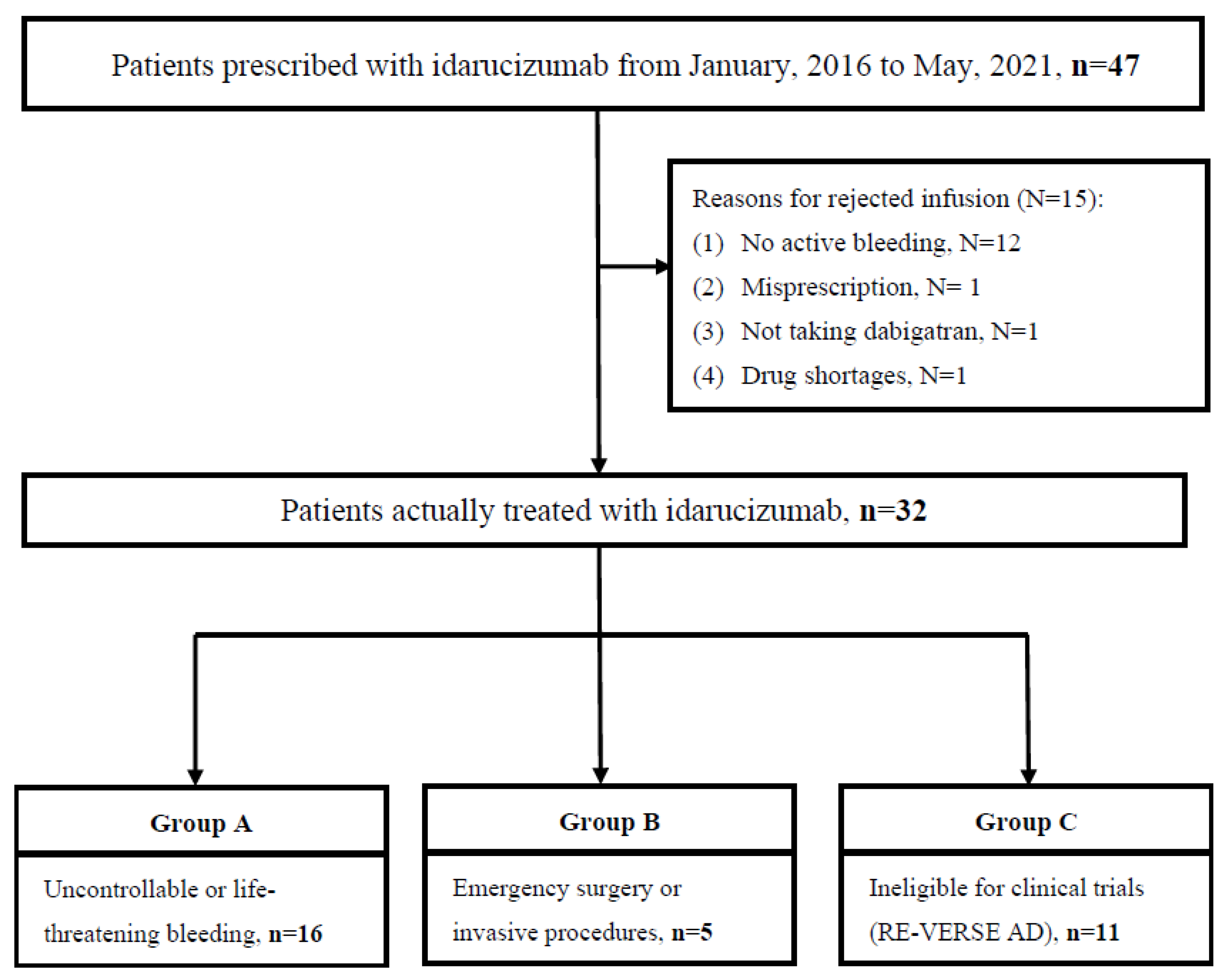
Medicina | Free Full-Text | Effectiveness and Safety of Dabigatran Reversal with Idarucizumab in the Taiwanese Population: A Comparison Based on Eligibility for Inclusion in Clinical Trials
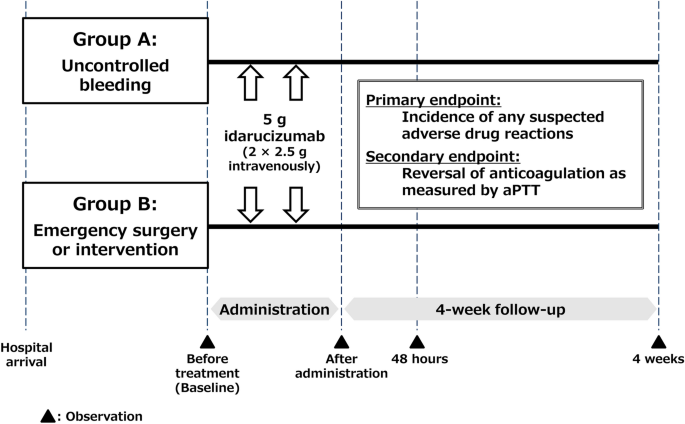
Idarucizumab for Emergency Reversal of Anticoagulant Effects of Dabigatran: Interim Results of a Japanese Post-Marketing Surveillance Study | SpringerLink

The Renal Elimination Pathways of the Dabigatran Reversal Agent Idarucizumab and its Impact on Dabigatran Elimination - Stephan Glund, Guanfa Gan, Viktoria Moschetti, Paul Reilly, Markus Honickel, Oliver Grottke, Joanne Van Ryn,